[ad_1]
Methods
Source of data and participants
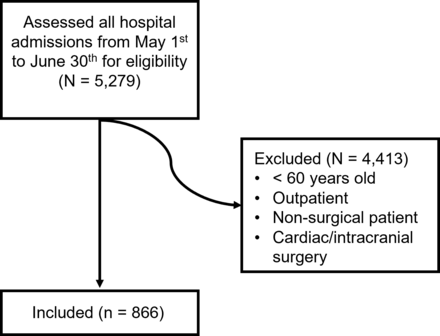
The delirium risk calculated using the PIPRA model was routinely collected, together with the delirium screening results, during a quality improvement project (QIP) in a Swiss, private 335-bed hospital.20 The project included all inpatients aged 60 and above undergoing non-cardiac, non-intracranial surgery with an admission date from 1 May to 30 June 2023. In 2023/2024, the hospital had a mix of 43.0% public and 57.0% private or partially privately insured patients, 79.4% of patients were outpatients and, of the inpatients, 82.4% were elective. The QIP aimed to increase delirium screening, treatment and prevention.
Comparison of validation to original development data
The development data originated from an IPDMA of clinical studies18 19 21 performed in several countries, whereas the validation data for this study consisted of real-world data collected as part of a QIP in a single private hospital in Switzerland. The eligibility criteria in this QIP were generally broader than those in the studies captured by the IPDMA. The delirium outcome was also measured heterogeneously in the IPDMA data, with only one study (contributing less than 10% of patients) that used the Delirium Observation Screening Scale (DOSS) as a diagnosis tool, although in combination with Diagnostic and Statistical Manual of Mental Disorders (DSM-IV) criteria. In the QIP presented here, the DOSS was used as the primary diagnostic tool, in combination with any routine diagnosis performed by the physician according to DSM-V criteria. In the development dataset, the predictors were collected specifically to investigate predictors of POD, while here they were collected during the routine preanaesthesia consultation.
Outcome
The outcome predicted by the model was POD, which was defined as delirium occurring up to 7 days after surgery. In this dataset, a patient was deemed delirious if they had a score of 3 points or more for the DOSS or had the ICD-10 diagnosis code for delirium for their hospital stay. The DOSS is a 13-point checklist for the identification of delirium.22 The data were collected in routine clinical practice, with delirium risk present in the electronic health record of the patient and viewable by the nurses performing the DOSS. The nurses were not informed about the validation project and were instructed to perform the DOSS on all patients as part of the QIP. Compliance with DOSS screening was assessed by comparing the observed number of DOSS screenings to the expected number. We anticipated one DOSS screening per patient per shift, with three shifts per day. Thus, compliance was calculated as a percentage:

Predictors
All PIPRA variables (age, body mass index, American Society of Anesthesiologists Physical Status Classification System (ASA) score, number of prescribed medications, cognitive impairment, history of delirium, surgery risk, laparotomy/thoracotomy, optional preoperative C reactive protein (CRP) value) except CRP were routinely collected. CRP is optional and therefore was only used where available.
Predictors were not collected specifically for the delirium risk. Instead, they were all part of clinical routine and were collected by the anaesthesiologist during the preanaesthesia consultation.
In addition to the main predictors, we recorded patient sex and the Self-Care Index (SPI) for further characterisation. The SPI, assessed daily, measures self-care abilities (eg, hygiene, mobility, elimination), producing a total score from 10 (maximum impairment) to 40 (full self-care).23
Sample size
A sample size was not calculated since it was a QIP. However, with a POD incidence of 10% and a targeted area under the curve (AUC) of 0.75, a 95% CI for the AUC would have a width of 0.12 with the 866 subjects enrolled in the study. We consider this to be precise enough to provide meaningful information about model performance.
Missing data
Missing data were only imputed for PIPRA predictors. Following the use proposed in the development paper, mean/mode imputation was used. When CRP was not available, the PIPRA submodel without CRP was used for prediction rather than imputing CRP, as in the development paper.
Statistical analysis methods
Subject and procedure characteristics were summarised by median (IQR) or frequency (percentage), dependent on data type. Differences in clinical parameters across groups (eg, POD/no POD) were explored using t-tests or χ2 tests, accordingly. All analyses were performed using R Statistical Software (V.4.3.2).24
This external validation was performed using data completely independent of the model development. To compute the predicted risk on the new data, the following equation was applied: p=1/(1+exp(−lp)), where p is the predicted risk and lp is the linear combination of the individual predictor variables multiplied by the log odds coefficients (including the intercept).
The participant and procedure characteristics were summarised and compared across the development and validation studies using the same methods as for comparing across POD. They were also visualised using violin plots or bar charts.
Both model discrimination (AUC) and calibration (calibration-in-the-large, calibration slope, calibration plot) were assessed on the validation data.
Risk groups
Patients were stratified into low (PIPRA <10%), intermediate (PIPRA from 10% to 19.9%), high (PIPRA from 20% to 34.9%) and very high (PIPRA >35%) risk groups as per the original publication.18 Patients with high risk or very high risk have more chance of developing delirium than the average elderly population. For this population, it is essential that some preventive perioperative measures are taken. An exploratory analysis was added, in which a stratified table was created based on risk group. The number of delirious patients was identified, and the number of potentially preventable cases was estimated, assuming prevention effectiveness rates reported in the literature.17
[ad_2]
Source link