[ad_1]
Results
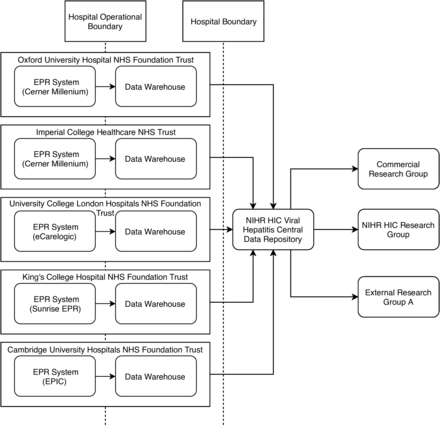
The NIHR HIC Viral Hepatitis Research Database has been developed and populated with data. Figure 3 provides an overview of the data model of the database and the relationships among different entities. The data collected falls into several categories: (a) basic information (eg, demographics, hospital visits, death, discharge and study sites), (b) laboratory data, (c) treatments, (d) diagnoses, (e) liver conditions, (f) hepatitis virus genotypes and (g) other clinical information.
Data model overview of the National Institute for Health Research Health Informatics Collaborative Viral Hepatitis Research Database. The defined dataset for viral hepatitis research includes 32 entities/tables, which can be grouped into different categories, marked in different colours. Each patient has a unique clinical data identifier that is stored in the table ‘clinical data IDs’. The relationship between two entities is shown beside their connection line. For example, 1:1 between table ‘clinical data IDs’ and table ‘study subject’ indicates that a patient can only have one study subject ID; while 1:N between table ‘clinical data IDs’ and table ‘hospital visit’ indicates that a patient could possibly have multiple hospital visits.
The database contains 32 tables for storing collected datapoints, and several tables for data field definitions. The final database consists of 349 data fields, split into 20 different element types. There are 203 data fields that are common to HBV, HDV, HCV and HEV. The remaining data fields are specific to a type of viral hepatitis (HBV and HDV (n=75), HCV (n=47) or HEV (n=24)). Due to the differences between sites, no datapoints were made mandatory. The current dataset was submitted between 7 December 2018 and 14 June 2019. The database contains 3494 patients with associated clinical information. There are 842 676 records regarding laboratory tests, 2824 records regarding imaging data and 8514 records for medications (including antiviral therapy and others), with data on comorbidities, diagnoses, genotype information, liver conditions, treatment side effects and social behaviour also included.
Chronic HBV cohort description
Data have been extracted from the NIHR HIC Viral Hepatitis Research Database by using HBV lab test information. Patients included in the extract meet at least one of the following criteria:
-
Two positive hepatitis B surface antigen tests, at least 6 months apart.
-
Positive hepatitis B surface antigen and positive HBV DNA, at least 6 months apart.
The characteristics of the chronic HBV cohort of 960 patients are described in table 2, including age, gender, race, HCV coinfection, HIV coinfection, comorbidities, the severity of liver disease and treatment information. Among these patients, 254 patients (26.5%) have previously received medications or are currently under treatment. There are 17 patients coinfected with HDV, with data extracted based on detectable HDV viremia or anti-HDV antibody. However, there were 475 patients who had no HDV test data. Missing data for each variable is reported as ‘unknown’ in table 2.
Characteristics of chronically infected hepatitis B virus (HBV) cohort
Chronic HCV cohort description
Data have been extracted for patients with a positive HCV RNA test or HCV genotype from the NIHR HIC Viral Hepatitis Research Database. The characteristics of HCV cohort of 1404 patients are described in table 3, including age, gender, ethnicity, HCV genotype, HBV coinfection, HIV coinfection, comorbidities, the severity of liver disease and treatment information. Among these patients, 914 patients (65.1%) had received interferon-free direct-acting antiviral treatment by February 2018. Similarly, to the HBV cohort, there were various degrees of missing data for each variable, which is reported as ‘unknown’ in table 3.
Characteristics of hepatitis C virus (HCV) cohort
HEV cohort description
Data have been extracted from the NIHR HIC Viral Hepatitis Research Database by using HEV IgM and HEV IgG lab test information. HEV usually causes an acute and self-limiting infection but can occasionally cause severe disease and/or chronic infection; our data collection approach sets out to include both categories. Patients included in the extract meet at least one of the following criteria:
-
Patients with anti-HEV IgM and anti-HEV IgG were both positive.
-
patients with HEV RNA detected in serum and/or stool.
There are 14 patients with acute HEV infection in our current database. Among them, 4 patients are male, and gender information was missing for the other 10. The median age at the time of patients first positive HEV test was 61.5 years (range: 21–86).
[ad_2]
Source link