[ad_1]
Abstract
Objectives While mobile health (mHealth) interventions are widespread, few studies assess impacts at the population level in low-income and middle-income countries. South Africa’s tuberculosis (TB) burden is high, and a substantial share of cases remain undiagnosed. We evaluate the impacts of community activations of TBCheck—a WhatsApp/USSD-based chatbot that allows individuals to evaluate themselves for TB risk.
Methods We use a quasi-experimental approach comparing treated and control subdistricts nationally before and after community activations using dashboard data from the TBCheck platform and weekly or quarterly subdistrict TB test data from the National Health Laboratory Service. Dependent variables are the number of self-screening tests on the platform, total tests and number of positive tests per subdistrict. We employ dynamic difference-in-difference models accounting for subdistrict unobservables and time trends using weekly data, and synthetic control methods matching on preintervention trends in outcomes using quarterly data.
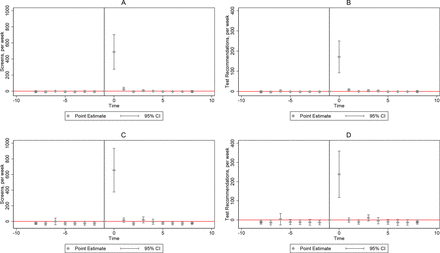
Results Impact estimates suggest an increase in the number of self-screening tests on the platform (487.53, p-value<0.01) as well as TB tests (107.90, p-value=0.05) in treated relative to control subdistricts due to intervention activities in the week of the intervention. After 2 weeks, impacts on the number of self-screening tests are insignificant (−6.18, p=0.23), and after 1 week, impacts on TB tests are insignificant (36.44, p-value=0.32).
Discussion and conclusion Activation activities associated with TBCheck led to short-lived and variable impacts on uptake and tests in target subdistricts. Alternative strategies are required for sustained uptake of such mHealth tools.
Introduction
Promotion of self-screening and early detection of communicable diseases is a priority for public health yet challenging due to behavioural complexity. Mobile health (mHealth) has been identified as an important solution. The term broadly collects mobile wireless technologies for public health, ranging from the basic provision of information and services through SMSes to more sophisticated smartphone applications.1 MHealth interventions have been used to promote screening and testing for COVID-19, TB and Ebola in African countries.2 Despite widespread application, little is known about the impacts of such applications on case identification at the population level. A recent systematic review of mHealth platforms for case identification in African countries reports that assessments tend to focus on the number of self-screen tests via mHealth platforms and do not consider the translation of positive self-screens to testing and case identification.2
This study contributes an in-depth, rigorous assessment of the TBCheck platform’s impact on self-administered screening for potential TB, but also on testing and case identification. TBCheck was developed to provide scalable, affordable support for post-COVID TB case detection in South Africa (SA). The country accounts for 3.6% of the global TB burden and is among the 14 countries with the highest burden of TB, TB/HIV and multidrug resistant TB.3 TB-related mortality is the leading cause of death in SA.4 5 The high rate of HIV coinfection continues to amplify both the spread and the mortality of the epidemic.6 SA’s 2018 TB prevalence survey found 852 bacteriologically confirmed pulmonary TB cases per 100 000 and a total of 150 000 ‘missing’ ones where people tested positive for TB in the survey but had not yet been tested or diagnosed and were therefore also not on treatment.7 8 Treatment delays are associated with a higher likelihood of hospital referral and mortality.9
The TBCheck platform was conceived as a response to ‘missing’ TB cases, as a way for people to screen themselves for possible infection and encourage those identified as high risk to get tested for TB. The tool was also envisaged as a response to the dramatic decline in TB testing during the pandemic. TBCheck followed the design of the HealthCheck COVID-19 self-assessment tool, which screened over 1 million people.10 The HealthCheck tool’s COVID self-screen tests were achieved when these were mandated for entry to government buildings to study and work, so it remains unclear if similar uptake will occur for TB where screening is not compulsory. Similar to HealthCheck, TBCheck is accessed through the National Department of Health’s WhatsApp and USSD chatbot support service. The TBCheck chatbot asked users five questions on their symptoms and risk of TB, and then classified users’ risk and, if indicated, advised them to test for TB. TBCheck was available nationally in March 2021 with relatively low take up, but in late 2022 and early 2023, week-long activation campaigns were undertaken in communities and clinics in eight subdistricts to promote awareness and use of the tool for self-screening.
This study aimed to assess whether these community activations promoting a mobile health TB screening platform could increase uptake of self-screening, as well as the number of TB tests and positive cases identified, and whether any increases would be sustained over time. The emphasis was on interventions that would be replicable and sustainable within the constraints of the public health system and budgets.
Discussion
We robustly documented that activation activities in subdistricts led to short-term increases in weekly TBCheck usage relative to control subdistricts as evidenced by the platform’s dashboard data. We also found that there were positive spillovers to neighbouring districts in the usage of TBCheck. Using NHLS data, we then tested if the increase in self-screenings translated to changes in weekly TB tests, positive tests, as well as the positivity rate. In line with the brief activity increases on TBCheck, we found a short-term effect on the number of tests but not on case findings nor the positivity rate. In other words, TBCheck promotion activities did not lead to sustained effects on TB testing beyond the initial week of activities nor on TB case detection.
Our study has limitations: (1) intervention subdistricts were not picked randomly for logistical and programmatic reasons. Our estimates, specifically in quarterly data, may thus be driven by dynamic confounders, including time-variant unobservables or other health programmes and shocks occurring at the same time, leading to violation of the parallel trends assumption. (2) Only a few heterogeneous subdistricts received intervention activities due to budget constraints. (3) Activation activities had low coverage and were of low and variable intensity, which may have contributed to the lack of sustained and large effects, and we may lack power to detect more subtle longer term effects. Effects were clearly concentrated in the short term and thus limited our ability to document heterogeneity and longer term effects using synthetic control models. Testing the effects of longer term interventions is a possible avenue for future research. (4) We presented evidence that activities spilled over to neighbouring areas, leading to benefit sharing. (5) We evaluate the impact of specific community activities and not the effects nor the specificity of the screening platform alone or mHealth in other contexts with different TB burdens and dynamics. (6) We lack information on intervention subdistricts that could further help to refine the analysis, including ‘missing TB cases’ and TB incidence. That said, such population trends are slow-moving, while we examine effects within weeks of a brief intervention. The subdistrict fixed effects absorb time-invariant confounders. (7) A final limitation of some of the intervention strategies is that individuals with active TB may feel unwell and may thus not be found in shopping centres. However, activities were therefore also covered in clinics.
Conclusion
Community promotion activities briefly boosted the usage of a mHealth platform for TB self-screening and translated into brief increases in TB testing. These effects were not sustained, and there was no increase in positive case detection. Longer-term approaches and more sustained interventions and investments would be needed to yield enduring and large increases in self-screening and case detection at the population level. While mobile health screening tools have the potential to reach large shares of the population at risk, uptake of such tools remains a major challenge to solve for practitioners and policy stakeholders. To accelerate learning, we need better research and assessment of existing platforms and tools, and in particular assessments including the impact of screens on testing and case identification.
[ad_2]
Source link