[ad_1]
Results
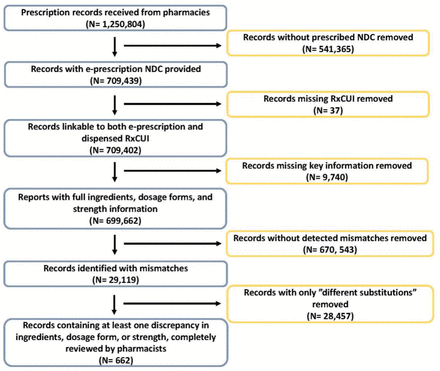
Of the 1 250 804 records sent for processing, 699 662 contained all the required information to compare the prescribed item with the dispensed, and 29 119 mismatches were reported. After cleaning, 662 records with complete pharmacist reviews remained, comprising 587 (88.7%) intended and 75 (11.3%) unintended mismatches (figure 1). The overall dispensing error rate was 0.01% (75/699,662).
Flow chart of e-prescription and dispensing medication record processing and analysis. NDC, National Drug Code; RxCUI, RxNorm concept unique identifiers.
One pharmacy was excluded due to incomplete pharmacist reviews. Among the remaining 13 pharmacies, 69% were independent pharmacies, with 67% operating for over a decade. Weekly prescription volumes ranged from 500 to 3000, with a median of 1543. All pharmacies had at least one full-time pharmacist, 77% also employed part-time pharmacists and 92% had full-time technicians. Approximately half had overlapping pharmacist shifts (16–35+ hours per week). Standard services included free home delivery (92%), medication therapy management (92%), care plan development (85%), home visits (48%), 24-hour emergency services (48%) and compounding services (62%). Additional pharmacy demographic details are provided in online supplemental appendix table 1.
Table 1 presents a basic comparison of intended and unintended mismatches across the included pharmacies. Median mismatches per pharmacy were higher for intended mismatches (32, IQR 14–52) than unintended mismatches (5, IQR 1–14). When comparing intended and unintended mismatches, significant differences were observed for different ingredients (26.2% vs 42.7%, p=0.004) and strengths (53.7% vs 36.0%, p=0.005). In comparison, dosage form differences were not statistically significant (47.4% vs 54.7%, p=0.27). Preferences for future alerts to the mismatches revealed a significant contrast, with 96.0% of respondents favouring alerts for unintended mismatches compared with 56.7% for intended mismatches (p<0.001).
Comparison of intended and unintended mismatches across pharmacies
As figure 2 shows, among intended mismatches, key reasons behind intended substitutions included prescriber-approved substitutions (366, 62.4%), dosage form changes (155, 26.4%), equivalent strength switches (124, 21.1%), stock issues (98, 16.7%), insurance or patient preferences (73, 12.4%) and other reasons (72, 12.3%). Multiple responses were permitted in a single record, with a median selection of 1 (IQR: 1–2, range: 0–4). The ‘other’ category overlapped with the previously mentioned factors. At the same time, additional causes included miscommunication or prescription errors, where prescribers had to clarify inconsistencies in strength or dosage forms and operational constraints, such as hospitals not completing prior authorisations, necessitating medication adjustments (eg, switching suspensions to capsules for g-tube use).
Reasons for intended mismatches between prescribed and dispensed medications
Regarding the 75 unintended mismatches, the majority of reported unintended mismatches (72, 96.0%) originated during the data entry, order or transcription stage. Most incidents (66, 88.0%) were categorised as events that reached the patient, while 9.3% were near misses and 2.7% were unsafe conditions. Among reported harm levels, pharmacists reported whether the patient sustained harm from the incident: 90.7% involved no harm, while 2.7% resulted in mild harm and 1.3% in moderate harm.
Figure 3 indicates that among unintended mismatches, contributors included human factors (62, 82.7%), drug name, label or packaging issues (57, 76.0%), unknown causes (23, 30.7%), workflow issues (12, 16.0%) and rare cases of staffing/scheduling or technology problems (1, 1.3% each). Multiple selections were also allowed in an individual record, with a median of 2 choices (IQR: 2–3, range: 0–3).
Contributors to unintended mismatches between prescribed and dispensed medications
Table 2 demonstrates the top 10 most frequent mismatches between prescribed and dispensed medications. Common discrepancies include formulation differences (eg, ophthalmic vs otic solutions), extended-release versus immediate-release formulations and variations in ingredient compositions and dosage strengths. Notable discrepancies include metformin hydrochloride 500 mg oral tablet being substituted with an extended-release tablet (n=21) and ofloxacin 3 mg/1 mL ophthalmic solution being dispensed as an otic solution (n=13). Other mismatches involve famotidine 10 mg oral tablet being replaced with a 20 mg strength (n=9) and albuterol sulfate 5 mg/1 mL inhalation solution substituted with a 0.83 mg/1 mL formulation (n=5).
Top 10 most frequent medication mismatches by ingredient, dosage form and strength
[ad_2]
Source link