[ad_1]
Results
The conceptualisation and purpose of CEBPWs
CEBPWs are diagnosis and therapy standards for diseases established by concretising large amounts of real-world clinical data through big data technology. CEBPWs are built on DIP groups and take into account individual patient characteristics and reflect real-world data characteristics.
They have two main objectives. The first is to regulate medical practices, reduce costs and improve the quality of medical services; the second is to establish a correlation system of payment standards, diagnostic standards and therapy standards by forming a CEBPW application model incorporating comparable value, assessable quality, controllable process and standardised operation among payment standards, diagnostic standards and therapy standards. The CEBPWs also serve as a benchmarking system for hospital management and healthcare insurance payment and supervision.
Key elements of CEBPWs
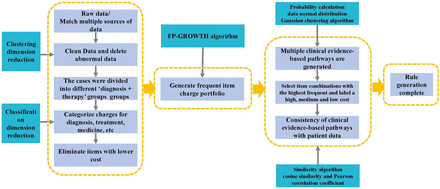
CEBPWs have five elements. The first key element is the identification of objective evidence—the use of real-world data features to concretise practical problems that need to be solved in clinical practice and management. The objective of developing CEBPWs is to integrate the diagnostic standards and therapy standards of the disease group with the DIP payment standards and match the cost payment with the diagnostic and therapy content of the disease group, including key diagnostic and therapy items and their structure, the order of item classification, item frequency, cost weight and time and sequence. It proceeds by clustering, classifying and summarising clinical data to yield data evidence. A variety of data evidence is collected to form evidence clusters, and logically related evidence is integrated into big data clinical evidence-based pathways around problems to be solved.
The second key element is the correct classification of evidence. The objective and reasonable classification of large amounts of chaotic clinically produced data can convert it into evidence. Evidence can be divided into two categories: diagnosis and therapy. Diagnosis items can be subdivided into imaging, examination, endoscopy, pathology, nuclear medicine, etc, and therapy items can be divided into surgery, anaesthesia, intervention, drugs and consumables. An evidence bank was established according to the classification of diagnosis and therapy items, and the evidence was integrated into CEBPWs for different disease groups according to different needs.
The third key element is evidence value weight. Different ‘diagnosis+therapy’ groups have different diagnosis and therapy methods, which involve a large number of diagnosis and therapy items. As the diagnosis and therapy items pose huge challenges to medical insurance payment and medical management, it is necessary to analyse the cost of each item based on data. According to a few key principles, the diagnosis of a DIP group accounts for 80% of the total examination cost, corresponding to 20% of the examination items, and these items were clustered and summarised as the evidence of diagnosis value weight. The evidence of therapy value weight is also formed in the same way for therapy items. These key core items must exist in the corresponding disease group to standardise the disease diagnosis and therapy process, ensure the quality of care and objectively fit the cost and medical technology level. Using weight of evidence value makes the formulation of CEBPWs simplified, and the use of data is more targeted and applicable.
The fourth key element is evidence frequency weight. In a ‘diagnosis+therapy’ group, different examination items have different probabilities of appearing; those with a high probability of appearing in the cases of the DIP group will be used to form frequency evidence through probabilistic data feature mining. High-frequency items are essential to the diagnosis and therapy process of the ‘diagnosis+therapy’ group and are of great significance to the management of diagnosis and therapy norms and the payment and supervision of health insurance. If they are missing, the quality of diagnosis and therapy will be affected. Evidence frequency weight can be analysed to identify missing items or frequency anomalies in clinical practice.
The fifth key element is temporal ordering evidence. Clinical diagnosis and therapy are in a strict chronological order, with a progressive relationship between diagnosis items and therapy items on a timeline. At the same time, each day of hospitalisation corresponds to different diagnosis items and therapy items. According to the diagnosis and therapy process, the temporal data characteristics of diagnosis items and therapy items are mined to form temporal evidence. The diagnosis and therapy process of each ‘diagnosis+therapy’ group is divided into three stages: diagnostic examination, treatment intervention and recovery observation, and different temporal evidence pertains to each stage, thereby standardising diagnosis and therapy behaviours and increasing their accuracy (figure 2).
CEBPWs of cardiac surgery and superimposed time dimension. CEBPWs, clinical evidence-based pathways.
Results of CEBPWs
We shall take orthopaedic musculoskeletal system surgery as an example. Criteria for the CEBPWs for musculoskeletal system surgery include a clinical laboratory test, clinical immunological test, clinical chemistry test, clinical haematology test and colour Doppler ultrasonography, histopathological examination and X-ray inspection, artificial joints, general anaesthetic, opiates, peripherally acting muscle relaxants, hypnotics and sedatives and other beta-lactam antimicrobials. The completion of musculoskeletal system surgery should include the key items mentioned above (figure 3). Each column displays the items of medical treatment, including diagnosis, therapy, drugs and consumable items. Based on diagnosis, therapy, drugs and consumable items, standardised pathways are generated for mandatory and high-frequency items, while different CEBPW pathways are generated based on the patient’s individual characteristics and needs.
CEBPWs of orthopaedic musculoskeletal system surgery. CEBPWs, clinical evidence-based pathways.
Application result
We established an evaluation and supervision system based on the CEBPWs. Through comparison of the CEBPWs of the DIPs and practice, an early warning judgement was formed and abnormal therapy modes and cost structure deviations were evaluated to implement precise hierarchical supervision at the institutional, departmental, doctor, patient group and case levels, where the smaller the granularity, the higher the classification precision, facilitating accurate supervision.
Next, we established a mechanism for detecting suspected violations. By combining the macro-operation of the medical insurance fund with the specific use of the fund, we have gradually constructed an intelligent monitoring model for medical insurance based on big data. Specifically, with regard to issues such as up-coding, cost deviation, disaggregated hospitalisation and cost transfer, a mechanism for the detection of suspected irregularities has been established based on the CEBPWs.
CEBPWs were implemented and applied in Shanghai, and 42 954 pathways were formed. In 2023, 79 hospitals (24 tertiary hospitals and 55 secondary hospitals) in 16 districts of Shanghai conducted self-inspections through CEBPWs and the DIP Intelligent Supervisory System. Violations were found in 433 cases, including 71 cases of upcoding, 106 cases of cost transfer, 117 cases of overtreatment and 139 cases of other violations, involving a total cost of ¥6.95 million. The item frequency deviation was 8.64% and the cost deviation was 10.82%, with 8.95% for diagnosis, 9.44% for therapy, 14.81% for drugs and 8.98% for consumables; cost overruns were 6.02% and negative values were 4.80%; and the chronological deviation was 39.63%. The deviation between CEBPWs and CPWs is relatively small, it indicates that CEBPWs include most of the items in the clinical pathway, and, therefore, the pathway generated by CEBPWs is reasonable.
[ad_2]
Source link