[ad_1]
Summary of findings
The case studies that we have explored described both the opportunities and challenges of using routine data in applied and implementation research. A key strength was the convenience of collecting data for effectiveness outcomes, health economic assessment and process evaluation, in one data extraction. Crucially for implementation research, routine data can assess the impact of a new intervention on a whole population. Working with an established database could streamline processes of recruitment, ethics and regulatory approvals, data collection.
Despite recognising the benefits, many researchers described their frustration with barriers that demonstrably hindered research timelines, delayed analyses and were perceived to require many weeks to be spent on inefficient bureaucratic processes. Our case studies illustrate months of wasted researcher time, costly extensions and frustrated primary care practices blocked from participating in research. Inconsistent coding was a challenge for researchers, and the need to develop ways to extract data from free text was highlighted as a priority as was understanding the impact of external influences on coding practice. The delay in accessing data was often a surprise to our patients and public contributors, who expressed frustration and disappointment at the multiple barriers preventing access to valuable information that could improve people’s lives.
Recommendations
We have collectively generated a list of five key recommendations to improve the efficiency of using primary care EHRs in research, as summarised in table 5.
Summary of key recommendations to improve the efficiency of using routine data for primary-care based research
The first three recommendations relate to the current devolution of R&D, governance and IT approvals to local organisations. This is in marked contrast to ethics approval which is granted nationally and is not reviewed or challenged locally. The ethics application was seen as a time-consuming but reasonable process with agreed timelines that ensured timely completion. In the context of governance and IT approvals for UK-wide studies, the need for multiple local applications with diverse requests for paperwork, hugely varied timelines and reaching different conclusions amounted to a ‘postcode lottery’ and was universally frustrating to the research teams. Agreed standard paperwork, self-populating from the IRAS application where possible, should ensure that standardised information is available locally to inform decisions.
Variable interpretations by data protection (and other information governance) officers of the GDPR implications for use of routine data were a particular challenge highlighted by the researchers. Any uncertainty raised by a data protection officer will be likely to push decisions towards an overly risk-averse position suggesting that improved training and mentorship could support less experienced or overly cautious officers. There is a need to raise awareness of the potential of data-enabled trials in the UK and disseminate successful case studies2 though knowledge of UK-wide positive approvals did not prevent individual data protection and local governance officers from disallowing willing practices from participating in the IMP2ART trial.
These issues were endorsed in a qualitative study by Mukherjee et al
9 in the context of creating a learning health system, concluding that there needed to be a streamlined governance process akin to the NHS ethics system. Standardised training for trial sponsors, data protection officers, local governance and IT governance officers and all who are involved with decisions about research use of routine data, might enable a decision taken once to be understood and accepted through the UK. Inefficiencies in the current governance processes place undue pressure on the local governance officers, as tasks are repeated unnecessarily and paperwork reapproved in multiple locations leading to slow responses and delays to recruitment timelines. This then impacts on funders as additional costs are requested to extend projects to accommodate the delays. The costs and resources associated with over-recruitment of practices that ultimately cannot be randomised (20% in the IMP2ART trial) mean that extensions may not always be possible within the original budget. We did not capture the perspective of practice staff whose willingness to participate in a nationally approved study was blocked by a local decision (or lack of decision) but is unlikely to have been a positive experience.
Preconsent is receiving attention as a solution for some of the challenges in the context of commercial trials,16 which might streamline patient recruitment via EHR databases. The Clinical Practice Research Datalink (CPRD) provides a precedent for use of routine data in practice-level research: individual GP practices consent to sharing pseudoanonymised records, and patients are informed via a Fair Processing Notice displayed in their practice waiting room and on the practice website of their right to opt-out at any time.17 This enabled the PLEASANT18 and TRAINS11 trials (table 1). Similarly, consent for low-risk and low-contact studies could be issued as an opt-out, with patients asked to opt-in to more sensitive research.
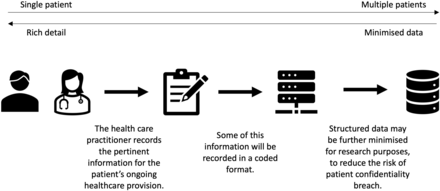
Clinical records are created to support the long-term provision of care, and decisions about suspected diagnosis and clinical management may be revised based on response to interventions and changes over time. Meaningful ascertainment of clinical features thus requires a review of records in series and application of some heuristic processes. This is possible with routine data but can be challenging. The use of different coding systems in primary and secondary care (eg, SNOMED-CT and the International Classification of Diseases, ICD-10) presents a specific challenge as while there are cross-maps between terms in different systems, the nuances may be lost in translation. Changes in coding practices over time owing to external events such as the COVID-19 pandemic or changes to the Quality and Outcomes Framework (QOF) must be understood so that there is continued validity to the inferred meaning of codes. These influences external to an individual study, not only impact the practical logistics of the trial and interpretation of findings but may affect the risk–benefit considerations that underpin an ethical trial design or may disrupt the evidence generated.
Finally, there is great scope for methodological research which can fulfil the dual aims of improving utility in routine clinical practice while generating high-quality research-ready data. Given the diversity of primary care clinical practice and the vast range of codes available, the choice of any consultation can be overwhelming. Enabling practice formularies that limit the initial choice without restricting the final selection of a nuanced code could simplify the process for clinicians while standardising the coding for secondary use. Clinical records tell ‘stories’ recorded as free text to inform future consultations. Progress in the use of natural language processing can enable relevant clinical information to be extracted (with appropriate IG safeguards) from the unstructured data in EHRs, which may improve the detection of symptoms and improve the performance of clinical prediction models.19 20 Semiautomated processes for extracting codes from free text as it is entered would improve the validity of inferences from coded data as the clinician would be prompted to reject inappropriate suggestions. This is particularly pertinent in the rapidly changing landscape of artificial intelligence; rapid evolution in governance guidelines is required to ensure that benefits can be achieved without compromising security.
Limitations and strengths
AUKCAR has links with the majority of UK applied researchers in asthma making it likely that we have included most large-scale research in this area. Together, we were able to comprehensively collate recurring issues preventing efficient research and provide detailed exemplars to describe the impact of these processes on publicly funded scientific research. Additionally, the AUKCAR collaboration includes researchers, healthcare providers and patients, and many members have multiple of these roles. As such, we have been able to thoroughly contemplate the conflicting needs and priorities of different parties.
This review voices the real-world experiences of the AUKCAR community throughout a series of large-scale studies, aiming to improve the lives of those living with asthma. The comprehensive 2022 review21 entitled ‘Better, Broader, Safer: Using Health Data for Research and Analysis’, by Goldacre and Morley presented 29 recommendations specifically related to information governance, as well as further recommendations in other domains. Many of these recommendations (particularly those under the subheading ‘Enhanced usability for IG and ethics processes’) are reinforced by the case studies we have described. For example, the seventh recommendation describes the objective to create a national centre for governance and regulations, which could develop standardised documentation and training, and provide top-level insights which would facilitate processes. In our current landscape, many people given ‘expert’ IG roles feel unprepared and understandably err on the side of caution.
Our study is limited to the UK research context (particularly pertinent, given the longevity of primary care EHR databases in the UK) though international literature has also highlighted similar issues,22 23 and the enablers and barriers identified have potential relevance to other countries using routinely collected data. Healthcare systems at an earlier stage of development of similar resources may be able to optimise the benefits and avoid the barriers that challenge our UK research. Similarly, we focused on asthma as an exemplar, but it is unlikely that experiences will be different in other disease areas (apart from COVID-19 when many of the regulatory processes were temporarily eased to enable a prompt response to the pandemic).
Finally, we note that while using routinely collected primary care data broadens the population compared with studies which specifically recruit participants, there are still some people not registered with, or not attending, a primary care practice, who will be missed in the population denominator. Linkage to other national or regional datasets may help detect the non-registered, but those who do not consult about their condition will still be missed.
[ad_2]
Source link