[ad_1]
Discussion
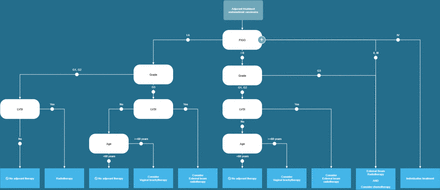
By combining real-world data with a computer-interpretable guideline, we successfully computed multiple aspects of adherence levels to the Dutch EC guideline. These results provide insights into guideline implementation by establishing a novel system designed to continuously evaluate adherence to guidelines, applied to EC.18 It establishes analytical standards within the context of guideline adherence, signifying the broader applicability of this approach beyond (endometrial) cancer.
The insights of adherence patterns within identified subpopulations serve as a cornerstone for discussing acceptable or optimal ranges of adherence by guideline committees and physicians.23 In general, one would reasonably expect that a specific subset of the patients is not treated according to the guideline, because in individual cases a guideline recommendation may result in more harm than benefit. In the current study, it was found that in two populations, a statistically significant increase in non-adherence occurred, warranting further investigation to understand the underlying phenomenon. Additionally, nearly 100% adherence was observed for adjuvant treatment in stage IA, grade 1 or 2 endometrioid carcinoma. In this case, the specific recommendation to refrain from further treatment likely accounts for the high adherence level. Optimal cut-off points for adherence will vary among different recommendations and subpopulations. Notably, anticipated adherence levels are likely correlated with the strength of the recommendation. Collaboration between clinicians, researchers and informaticians, supported by guideline adherence monitoring, is essential for enhancing these insights.
Significant variation was seen in primary treatment for stage I serous and clear cell carcinoma patients. The recommended intervention was implemented in 60% of cases; however, the remainder of the cases was distributed over 10 alternative types of interventions. We argue that this might be an indication for the guideline committee to reconsider this recommendation. In this example, the oncological outcome of the different interventions is probably quite similar, since in most of the interventions the uterus is surgically removed. In certain cases, the surgical procedures were quite extensive, as these most likely involved the diagnosis and treatment of ovarian cancer simultaneously. Ultimately, the diversity in surgical techniques and approach might result in differences in recovery, complications and long-term morbidity. Incorporating data on such outcomes would make guideline evaluation even more valuable.
The implications of our findings cascade into the broader paradigm of clinical practice guideline evaluation. Notably, our results spotlight the potential for continuous evaluation measurements as a tool for identifying areas of improvement and monitoring temporal shifts. Depending on the available data, many more analyses can be carried out, such as identifying differences per hospital (type), region, risk factors, multidisciplinary team discussion, trial participation, etc. This results in a meaningful clinical effect when acting steered by insights from daily practice. The practical application of such insights hinges on the harmonisation and technical standardisation of guideline knowledge and real-world data.24 As suggested by the results, a straightforward example is that paying close attention to providing accurate and as specific as possible descriptions of interventions in guidelines would greatly facilitate computable analyses. Ultimately, these developments contribute to the formation of learning healthcare systems.25
While our study and methodology yield substantial insights, it is important to acknowledge its limitations. The use of registry data introduces a temporal lag in obtaining adherence insights, caused by the delay in data availability to the registry. However, the turnaround time for this project using real-world data is significantly faster than conducting a clinical comparative study. Furthermore, our interpretation of non-adherence remains cautious, recognising that deliberate deviations from guidelines can be prudent and context-dependent. Yet, motivations underlying such deviations are often missing in the NCR dataset, and perhaps also in clinical reports. It would be highly interesting to register the reason for non-adherence to guidelines to gain more insight into this phenomenon. Finally, although the NCR dataset is extensive, its emphasis on the initial treatment phase restricts the range of evaluable clinical decision points outlined in the guideline. Expansion of or linkage with, for example, recurrence or clinical and patient-reported outcome data will greatly enhance the value of this type of evaluation.
Finally, the reliance on guidelines on randomised controlled trials (RCTs) often results in the exclusion of relevant subsets of patients.26 However, our study introduces a novel dimension, where variables governing RCT inclusion and exclusion, when present in real-world data, empower tailored and ongoing evaluation analyses. The implications of this approach are important, facilitating an inclusive evaluation of guideline adherence across varied patient profiles. This approach facilitates the identification of clinically relevant subpopulations that may benefit from a recommendation, adjusted to their specific characteristics. In addition, important developments are taking place in oncology in the field of, among other things, molecular diagnostics. It is likely that (endometrial) cancer subpopulations will become smaller as a result, and ongoing monitoring of guideline adherence using real world data will become increasingly important for revealing optimal interventions.27 Particularly when international collaboration can be established in the area of standardised computable guidelines and (real-world) data.
[ad_2]
Source link